Service Overview
The panel based on the clear target refers to the experimental method based on the molecular level and the cellular level, using the microplate form as the experimental tool carrier, using the automated operating system to execute the experimental process, and collecting the experimental result data with sensitive and fast detection instruments. The computer analyzes and processes the experimental data, detects tens of millions of samples at the same time, and supports the technical system of the whole system with the corresponding database.
In vitro high-throughput drug candidate screening is a core step in the preclinical R&D of antitumor drugs. The industry currently faces two major challenges:
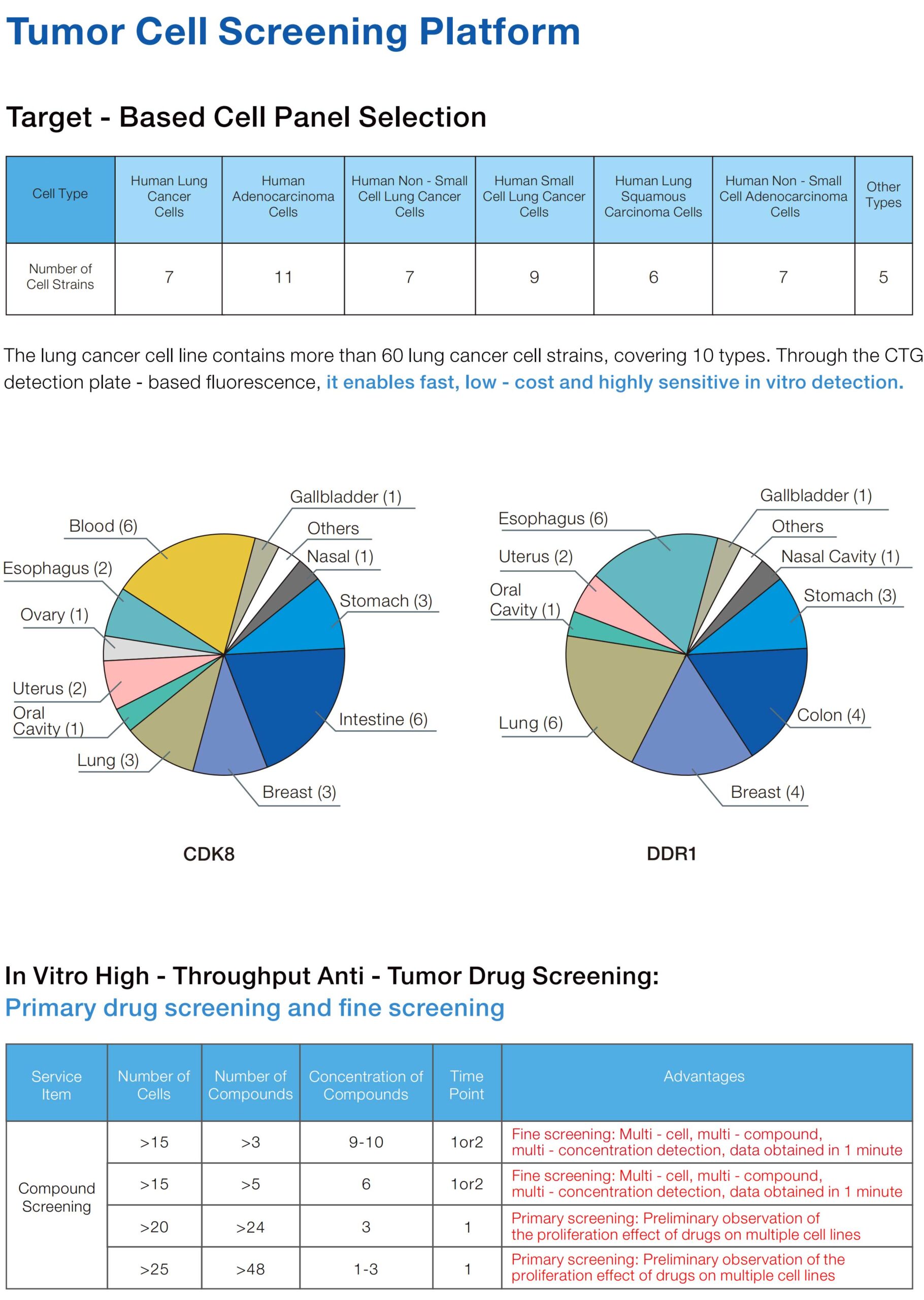
| Cancer Type | Number of Cell Lines | Detection Modules | Application Scenarios |
| Lung Cancer | 81 strains | Proliferation inhibition dose-response (CCK-8/CTG), Migration (Transwell) | Screening of targeted drugs/immunotherapy combinations |
| Colorectal Cancer | 42 strains | Proliferation inhibition dose-response (CCK-8/CTG), Colony formation (soft agar), Apoptosis (flow cytometry) | Research on chemoresistance mechanisms |
| Liver Cancer | 38 strains | Proliferation inhibition dose-response (CCK-8/CTG), Metabolism (OCR/ECAR), Invasion (Matrigel) | Evaluation of anti-angiogenic drugs |
Case 1: Screening with Lung Cancer Targeted Drug Panel
Case 2: Screening with Liver Cancer Combination Therapy Panel
For more cases: [link to the company case library]
Q1: Is customization of panels for rare cancer types supported?
A: Relying on the Chinese Primary Tumor Cell Bank (more than 100 types), panels for thyroid cancer, gallbladder cancer and other rare cancer types can be constructed.
Q2: Can the data delivery cycle be shortened?
A: Expedited service is supported, and cell proliferation testing can be delivered in as fast as 10 working days (30% additional fee required).
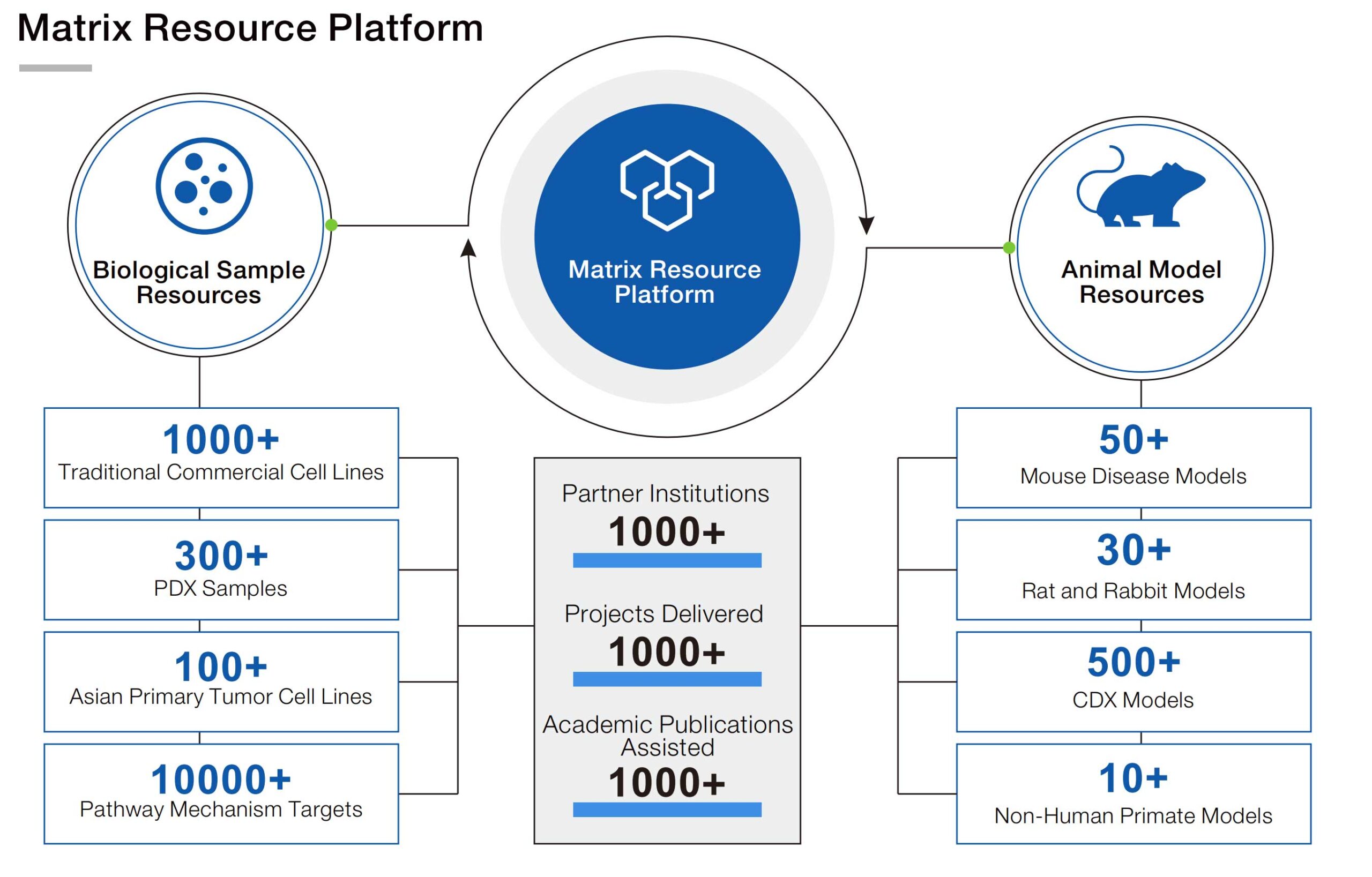
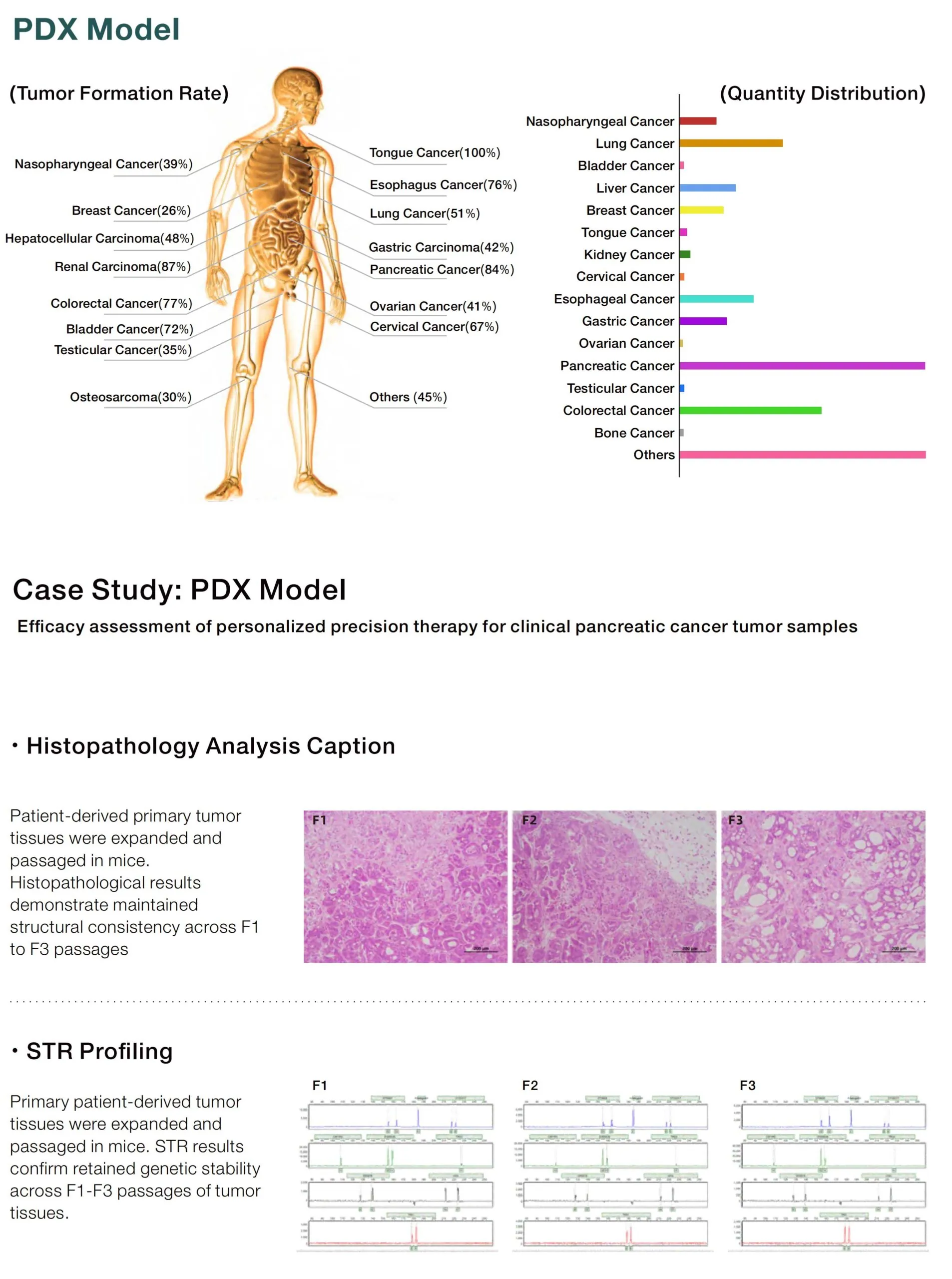
Get your exclusive solution now → [Request a Free Consultation]
Share details of your inquiry, and we’ll contact you shortly.
Room 238, Building A, Guangzheng Science and Technology Park, No. 11 Nanyunwu Road, Huangpu District, Guangzhou, Guangdong Province, China
3691125803@qq.com
+86 18802035152


Contact us via WeChat

Follow our Official Account